|
The desirable contrast in colour can be produced by scratch brushing with a pumice paste, or by use of a “greaseless” polishing compound on a buffing wheel. Modifications of these formulas called for the use of sodium, potassium, barium, and ammonium sulfides, which were claimed to produce different shades, but almost all sulfide colours are now produced from solutions of polysulfides which are sold in concentrated form under a number of trade names, usually called “oxidizing liquid.”Īll sulfide films require wet or dry scratch brushing for good appearance and will look better longer if protected by oiling, waxing or, more permanently, by a good top coat of clear lacquer. Originally liver of sulfur was employed, this being a crude mixture of potassium polysulfides and thiosulfate, also called potassium sulfuret. The use of chemical solutions is generally termed “oxidizing,” although the oldest method and the one which produces the widest range of brown to black stages on copper alloys actually produces not an oxide but a metal sulfide finish by the use of alkaline sulfide solutions. In conversion coatings, the metal surface is either converted into a protective film, usually, an oxide or sulfide of the metal involved, or a compound is precipitated which forms a surface film. Statuary finishes are conversion coatings. This datasheet presents the technology which underlies the craftsmanship and art involved in producing these coloured finishes. Chemical colouring techniques depend upon time, temperature, surface preparation, the mineral content of the water, humidity and other variables which influence the ultimate result. 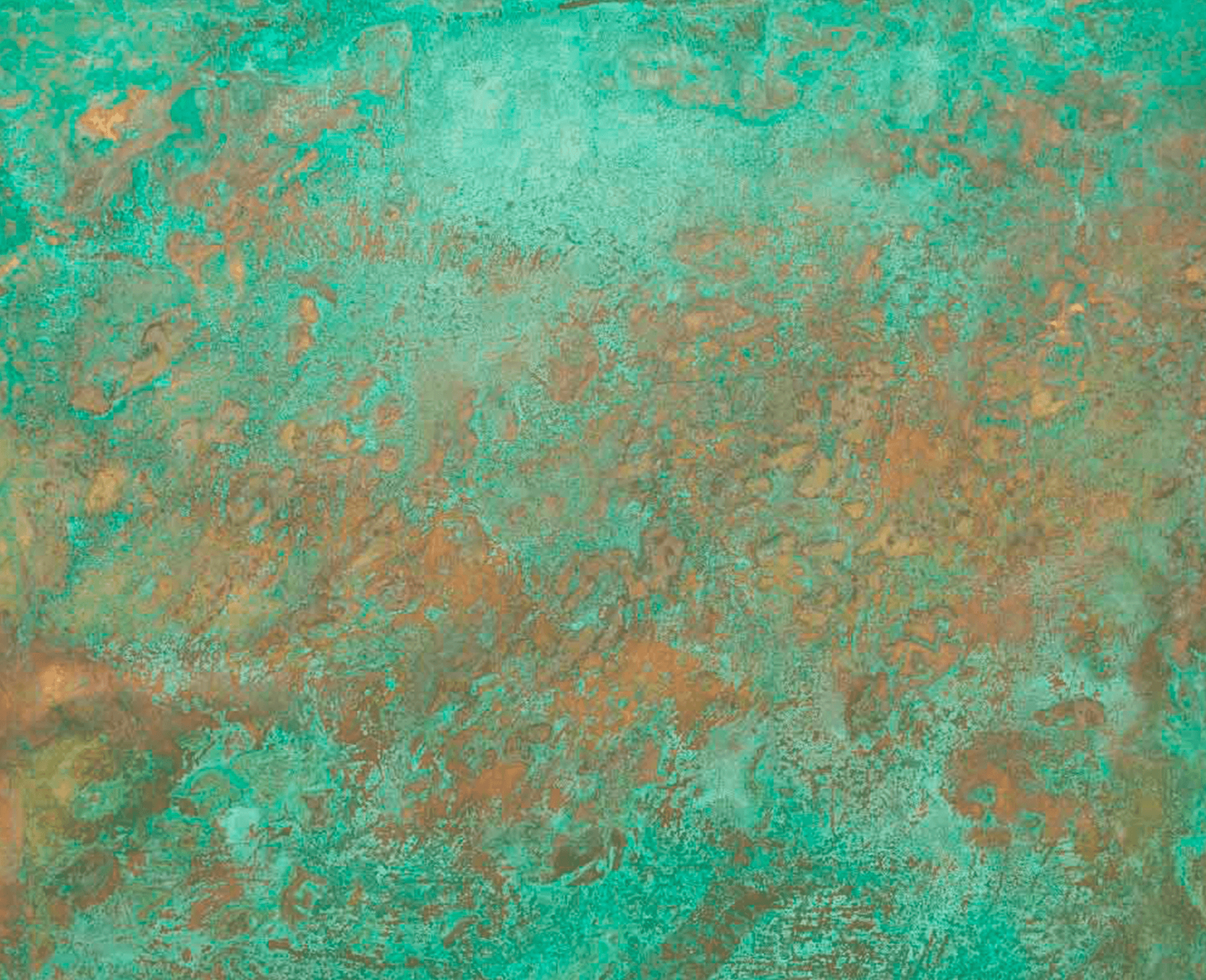
The wide range of colours and shades which may be achieved are largely a matter of craftsmanship and experience. While the chemical solutions described are those generally accepted in the metal finishing trade, many variations exist. This data sheet outlines procedures and formulations for producing both. The most common colours to be produced are referred to as brown or statuary finishes for bronze and green or patina finish for copper. While these metals may be used in their natural colour, as fabricated, it is sometimes desirable to chemically colour pure copper (C11000*), commercial bronze (C22000), architectural bronze (C38500) or other alloys referred to as “bronze” in architectural parlance. Table of ContentsĬopper and copper alloys are widely used in architectural applications to take advantage of their inherent range of colours. The limited availability for customers seeking aged, distressed & patinated finishes are not from a lack of demand but rather ability.This Data Sheet for Applying Statuary and Patina Finish to Copper is published by the Copper Development Association and has been provided by Austral Wright Metals. Lately, the focus has been mainly on margin & equipment to move the industry rather than developing better skill-based methods. It relies upon industry standards like single or two-coat systems as common methods to produce metal coatings. The current culture around powder coating doesn’t lend itself easily to visual innovation. They are bound by this rule or risk losing the quality of the effect they are trying to create if not heeded. 
It was my muse and yet it’s a painter’s crux. 
Speed in production using industrial powder coatings as a creative force intrigued me the most. 
It was my unique background as a master painter that challenged me to capitalize and create this finish. We call it perfect patina in paradise and it’s just one of the many effects created. I’m introducing a new twist to achieving a realistic patina copper effect on metal in a non-traditional way.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
